 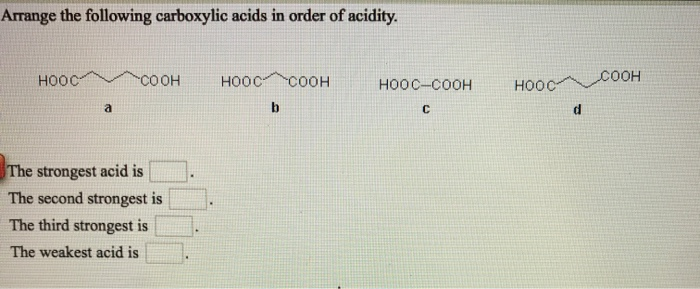
So because of that, um, the one that is directly connected of the ring is going to be stronger than the one that is isolated from the ring, So this will be the weakest often. Um, the lone pair left over on this alcohol on this oxygen would not be able to be stabilized by the ring because they can't reach it. Because there's this methylene group in the way here. The lone pair that would be left over if this hydrogen were removed is able to be stabilized by the residents in the ring while on this compound. And then we have these two alcohols leftover to compare so on Pinole, this first compound right here because the alcohol is directly attached to the benzene ring. So that makes them very strong assets on. 
And that's because residents with the carbon eel is able to stabilize the lone pair left over on the oxygen when the proton is removed. And now, in the chapter, we see the reason why carve oxalic acids are so much stronger than alcohols. So rough about we know that carb oxalic acids are stronger acids and alcohol's, so this is automatically our strongest asset. And then this last one, we have a car box to look acids that will be our acidic hydrogen. Our acidic hydrogen is here in this first one. This question asked us to rank the acidity of each of these compounds from strongest to weakest. And that's the answer to Chapter 19 problem number 27. We're going to decrease the acidity of the carb, oxalic acid and electron withdrawing substitutes are going to increase the acidity of the car box of gases. Electron withdrawing s Oh, this is going to be our most acidic molecule by four Here, Um, and so again, to solve this, we just need to remember the rules. Ah, and then the tri Floro metal group eyes strongly. I should have said least acidic intermediate acidity here because we don't have electron donation or electron withdrawal. 
I said the Benz Oic acid here, that's going to be our intermediate Oops. Um, the un substituted for I guess the the aromatic ring where only the only substitute is the carb oxalic acid. Transcribed Image Text: Arrange the following carboxylic acids in order of acidity. It acts as an electron donating group, and so decreases the acidity of the carb. So, um, are least acidic is going to be, uh, this third molecule with the metal group. Oxalic acid and electron withdrawing groups are going to increase the acidity of the car back. Um And so electron donating groups on the aromatic bring are going to decrease the acidity of the carb. Um, but so all three of these are aromatic carb oxalic acids, and so we need to remember our rules for the way that this works. Um, and so I always like to start by redrawing these, Um, I think it makes it easier to work with if they're, like, line structures instead of bone sticks structures. Three car box, sell gas IDs, and it asks us to rank them from least acidic to most acidic. Ah, and this problem gives us three ball and stick models of molecules. Problem number 27 from the Smith Organic Chemistry textbook. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
